|
ANALYSIS OF CARBOHYDRATES7. Analysis of Carbohydrates. Introduction. Carbohydrates are one of the most important components in many foods. Individual molecules can be. Some carbohydrates are digestible by. Indigestible carbohydrates. Consumption of significant quantities of dietary fiber has. 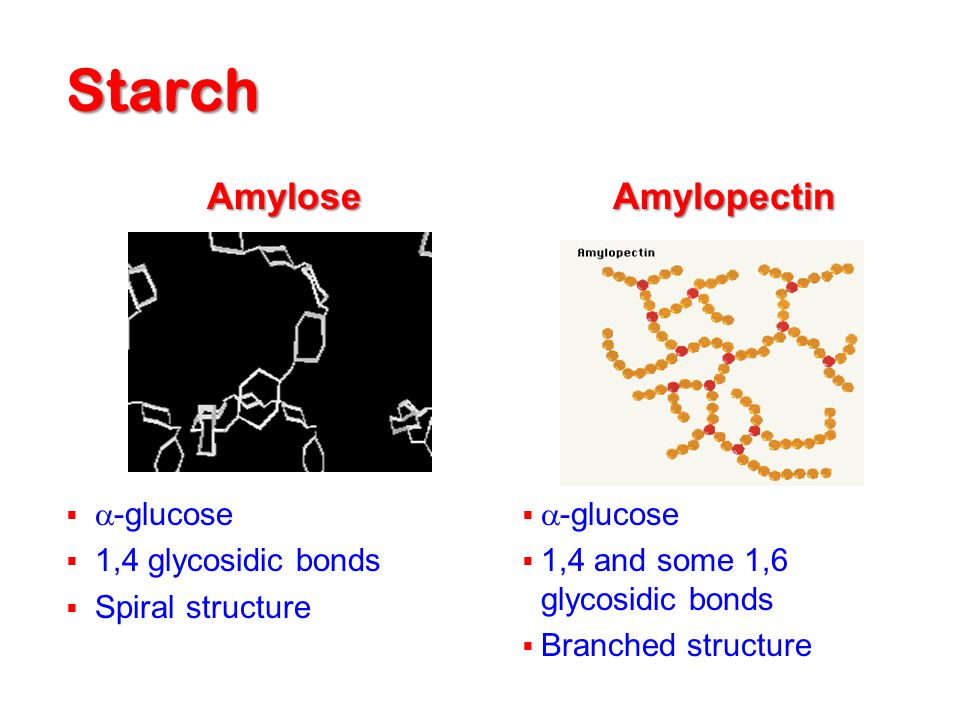
Glycosidic linkage named after the anomers of monosaccharide units (alpha and beta units) involve in this linkage. The position of alpha and beta glycosidic bonds can. UNESCO – EOLSS SAMPLE CHAPTERS CULTIVATED PLANTS, PRIMARILY AS FOOD SOURCES – Vol. I – Starch Bearing Crops as Food Sources - Krisztina. Study Campbell Biology: Chapter 5 Test Preparation flashcards taken from chapter 5 of the book Campbell Biology. Carbohydrates - Chemical Structure Disaccharides consist of two simple sugars. Sucrose, also called saccharose, is ordinary table sugar refined from sugar cane or. Study online flashcards and notes for Biology Textbook.pdf including LibraryPirate Interactive Figures Vivid images with related questions help you come to class. As. well as being an important source of energy and dietary fiber, carbohydrates. It is important to determine the. Standards of Identity. Nutritional Labeling - to. Detection of. Adulteration - each food type has a carbohydrate. They are aliphatic aldehydes or ketones which contain one carbonyl group and one or more. Most natural monosachharides have. Commonly occurring hexoses in foods are. The reactive centers of monosaccharides are the carbonyl and hydroxyl groups. Oligosaccharides. These are relatively low molecular weight polymers of monosaccharides. Disaccharides consist of two monomers, whereas trisaccharides. Oligosaccharides containing glucose, fructose and galactose monomers are the most commonly occurring in. Polysaccharides. The majority of carbohydrates found in nature are present as.   
Polysaccharides are high molecular weight polymers of monosaccharides (> 2. Polysaccharides containing all. The carbohydrate content of a food can be determined by calculating. Nevertheless. this method can lead to erroneous results due to experimental errors in any of. Sample Preparation. The amount of preparation needed to prepare a sample for carbohydrate. Aqueous solutions. On the other hand, many foods contain carbohydrates that are. In these foods it is usually necessary. The precise method of carbohydrate isolation depends on the. For example, foods are usually dried under vacuum (to prevent. Carbohydrates are polyhydroxy aldehydes, ketones, alcohols, acids, their simple derivatives and their polymers having linkages of the acetal type. They may be. INTRODUCTION. 
One of the most commonly used methods of extracting low molecular weight. Monosaccharides and oligosaccharides are. The soluble components can be separated from the insoluble. These two fractions can then be dried and. In addition, to monosaccharides. It is usually. necessary to remove these components prior to carrying out a carbohydrate. This is commonly achieved by treating the solution with clarifying. Clarifying agents. For this reason solutions are usually clarified. The most commonly used clarifying agents are heavy. However, it is important that the clarifying agent does. Ion- exchange. Many monosaccharides and oligosaccharides are polar. By using a. combination of a positively and a negatively charged column it is possible. Non- polar molecules can be removed by. Chromatographic and Electrophoretic. Chromatographic methods are the most powerful analytical techniques for the. Thin layer chromatography (TLC), Gas. GC) and High Performance Liquid chromatography (HPLC) are. Carbohydrates are. Carbohydrates can be. HPLC is currently the most important. In addition, GC requires. HPLC samples can often be analyzed. HPLC and GC are commonly used in conjunction with NMR or mass. Carbohydrates can also be separated by electrophoresis after they have been derivitized to make them electrically charged, e. A solution of the derivitized. The. carbohydrates are then separated on the basis of their size: the smaller the. Chemical methods A number of chemical methods used to determine monosaccharides. The concentration of carbohydrate. Non- reducing carbohydrates can be determined using the same. It is possible to. Many different chemical methods are. Most of these can be divided into. An example of each of these different types is given below. Titration Methods The Lane- Eynon method is an example of a tritration method of determining the concentration of. A burette is used to add the carbohydrate solution. The reducing. sugars in the carbohydrate solution react with the copper sulfate present in. Once all the copper sulfate in solution has reacted, any further. The. reaction is not stoichemetric, which means that it is. The disadvantages of this method are (i) the. Gravimetric Methods. The Munson and Walker method is an example of a gravimetric method of. Carbohydrates are. The concentration of precipitate present. This method suffers. Lane- Eynon method. Colorimetric Methods. The Anthrone method is an example of a. Sugars react with the anthrone reagent under. The sample is mixed with. The solution is then allowed to cool. There is a linear relationship. This method determines both reducing and non- reducing sugars because of. Like the other methods it. The Phenol - Sulfuric Acid method is an example of a colorimetric method. A clear aqueous solution of the carbohydrates to be analyzed. The solution turns a yellow- orange color as a result of the. The absorbance at 4. The. sulfuric acid causes all non- reducing sugars to be converted to reducing. This method is. non- stoichemetric and so it is necessary to prepare a. Enzymatic Methods. Analytical methods based on enzymes rely on their ability to catalyze. These methods are rapid, highly specific and sensitive to. In addition, little sample preparation is usually required. Liquid. foods can be tested directly, whereas solid foods have to be dissolved in water. There are many enzyme assay kits which can be purchased commercially to. Manufacturers of these kits provide. The two methods most. Some examples of the use of enzyme methods to determine sugar. D- Glucose/D- Fructose. This method uses a series of steps to determine the concentration of both. First, glucose is converted to. G6. P) by the enzyme hexakinase. ATP. Then, G6. P is oxidized by NADP+ in the presence of. G6. P- dehydrogenase (G6. P- DH). The fructose concentration is then determined by converting the. Maltose/Sucrose. The concentration of maltose and sucrose (disaccharides) in a sample can be. The maltose and sucrose are. The major problem with this method is that many other. Various other enzymatic methods. Food Analysis Nielssen). Physical Methods. Many different physical methods have been used to determine the carbohydrate. These methods rely on their being a change in some. Commonly used methods include polarimetry. IR, and density. Polarimetry. Molecules that contain an asymmetric carbon atom have the ability to rotate. A polarimeter is a device that. A polarimeter consists of a source of. The extent of polarization is. A calibration curve of a versus concentration is prepared using a. The concentration of carbohydrate in an. Refractive Index. The refractive index (n) of a. In practice, the. The RI is also temperature. C) and wavelength. This method is quick and simple to carry out and can be performed. It is used routinely in industry to. Density. The density of a material is its mass divided by its volume. The density of. aqueous solutions increases as the carbohydrate concentration increases. Thus. the carbohydrate concentration can be determined by measuring density, e. This technique is routinely used in. Infrared. A material absorbs infrared due to vibration or rotation of molecular. Carbohydrates contain molecular groups that absorb infrared radiation. By carrying out measurements at a number of. Measurements are. Analytical instruments based on infrared absorbance are. More sophisticated instrumental methods are capable of providing information. NMR or mass spectrometry. Immunoassays. Immuoassays are finding increasing use in the food. With time the animal develops antibodies specific for the. These antibodies can then be extracted from the animal. Immuoassays are. extremely sensitive, specific, easy to use and rapid. Analysis of Polysaccharides and Fiber. A wide variety of polysaccharides occur in foods. Most polysaccharides contain somewhere between 1. Some polysaccharides. Some polysaccharides exist as. Some polysaccharides. Consumption of many types of dietary fiber has been shown to have. Analysis of Starch. Starch is the most common digestible. In its natural form starch exists as water- insoluble granules (3 - 6. These two. kinds of starch have different physiochemical properties and so it is often. Sample preparation. The starch content of most foods cannot be determined directly because the. It is therefore necessary to isolate starch from. In natural foods, such as legumes, cereals or. Processed food samples are normally dried, ground and then dispersed. The monosaccharides and. Hence, the starch can be separated from the sugars by filtering or. If any semi- crystalline starch is present, the. C). Addition of perchloric acid or calcium chloride to the water prior to. Analysis methods. Once the starch has. Specific enzymes are added to. The glucose. concentration is then analyzed using methods described previously (e. The starch concentration is. Iodine can be added to the. If there are no other. The amylose and amylopectin concentrations in a sample can be determined. This can be. achieved by adding chemicals that form an insoluble complex with one of the. Some. of the methods mentioned will not determine the concentration of resistant. Analysis of Fibers. Over the past twenty years or. Adequate intake of dietary fiber is therefore. Dietary fiber is defined as plant polysaccharides. The major components of dietary. Some types of starch, known as resistant starch. The. basis of many fiber analysis techniques is therefore to develop a procedure. Major Components of Dietary Fiber Cell Wall Polysaccharides. Cellulose occurs in all plants as the principal. The type and extent of these associations determines. Cellulose molecules. Hemicelluloses are a heterogeneous group of. By definition. hemicelluloses are soluble in dilute alkali solutions, but insoluble in water. Pectins are another form of heteropolysaccharides. Non Cell Wall Polysaccharides. This group of substances are. Non- cell wall polysaccharides include hydrocolloids such as guar and. Lignin. Lignin is a non- carbohydrate polymer that. It is. usually associated with cellulose and hemicelluloses in plant cell- walls. Common Procedures in Sample Preparation and Analysis. There are a number of procedures that are. Lipid removal. The. Protein removal. Proteins. The resulting amino acids. Starch removal. Semi- crystalline. The glucose is then. Selective precipitation. Dietary fibers can be separated from other components in.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
August 2017
Categories |
 RSS Feed
RSS Feed
